Datasheet and periodic table PDF

| Title | Datasheet and periodic table |
|---|---|
| Author | Christina Sun |
| Course | Chemistry 1B |
| Institution | University of Sydney |
| Pages | 4 |
| File Size | 218.9 KB |
| File Type | |
| Total Downloads | 6 |
| Total Views | 139 |
Summary
Datasheet and periodic table
CHEM1112 learning material...
Description
DATA SHEET Physical constants Avogadro constant, Faraday constant, Planck constant,
NA = 6.022 1023 mol–1 F = 96485 C mol–1 h = 6.626 10–34 J s
Speed of light in vacuum, Rydberg constant,
c = 2.998 108 m s–1 ER = 2.18 10–18 J
Boltzmann constant, Permittivity of a vacuum, Gas constant, Charge of electron,
kB = 1.381 10–23 J K–1 0 = 8.854 10–12 C2 J–1 m–1 R = 8.314 J K–1 mol–1 = 0.08206 L atm K–1 mol–1 e = 1.602 10–19 C
Mass of electron, Mass of proton, Mass of neutron,
me = 9.1094 10–31 kg mp = 1.6726 10–27 kg mn = 1.6749 10–27 kg
Properties of matter Volume of 1 mole of ideal gas at 1 atm and 25 C = 24.5 L Volume of 1 mole of ideal gas at 1 atm and 0 C = 22.4 L Density of water at 298 K = 0.997 g cm–3 Specific heat capacity of liquid water = 4.181 kJ / kg °C
Conversion factors 1 atm = 760 mmHg = 101.3 kPa = 1.013 bar
1 Ci = 3.70 1010 Bq
0 C = 273 K 1 L = 10–3 m3 1 Å = 10–10 m 1 eV = 1.602 10–19 J
1 Hz = 1 s–1 1 tonne = 103 kg 1 W = 1 J s–1 1 J = 1 kg m2 s–2
Decimal fractions Fraction Prefix Symbol 10–3 milli m 10–6 micro 10 10–12 –9
nano pico
n p
Decimal multiples Multiple Prefix Symbol 103 kilo k 106
mega
M
109 1012
giga tera
G T
Standard Reduction Potentials, E Reaction S2O82−(aq) + 2e− → 2SO42−(aq) Co3+(aq) + e– → Co2+(aq) Ce4+(aq) + e– → Ce3+(aq) Au+(aq) + e– → Au(aq) Pb4+(aq) + 2e– → Pb2+(aq) MnO4–(aq) + 8H+(aq) + 5e– → Mn2+(aq) + 4H2O Au3+(aq) + 3e– → Au(s) Cl2 + 2e– → 2Cl–(aq) O2 + 4H+(aq) + 4e– → 2H2O Pt2+(aq) + 2e– → Pt(s) Br2 + 2e– → 2Br–(aq) MnO2(s) + 4H+(aq) + e– → Mn3+ + 2H2O NO3–(aq) + 4H+(aq) + 3e– → NO(g) + 2H2O Pd2+(aq) + 2e– → Pd(s) NO3–(aq) + 10H+(aq) + 8e– → NH4+ (aq) + 3H2O Ag+(aq) + e– → Ag(s) NO3−(aq) + 2H+ + e− → NO2(g) + H2O Fe3+(aq) + e– → Fe2+(aq) I2 + 2e– → 2I– (aq) Cu+(aq) + e– → Cu(s) O2(g) + 2H2O + 4 e− → 4 OH−(aq) Cu2+(aq) + 2e– → Cu(s) Sn4+(aq) + 2e– → Sn2+(aq) 2H+(aq) + 2e– → H2(g) Fe3+(aq) + 3e– → Fe(s) Pb2+(aq) + 2e– → Pb(s) Sn2+(aq) + 2e– → Sn(s) Ni2+(aq) + 2e– → Ni(s) Co2+(aq) + 2e– → Co(s) Cd2+(aq) + 2e– → Cd(s) 2CO2(g) + 2H+(aq) –+ 2e– → H2C2O4(aq) Fe2+(aq) + 2e– → Fe(s) Cr3+(aq) + 3e– → Cr(s) Zn2+(aq) + 2e– → Zn(s) 2H2O + 2e– → H2(g) + 2OH–(aq) Cr2+(aq) + 2e– → Cr(s) N2(g) + 4H2O + 4 e− → N2H4(aq) + 4 OH−(aq) Al3+(aq) + 3e– → Al(s) Mg2+(aq) + 2e– → Mg(s) Na+(aq) + e– → Na(s) Ca2+(aq) + 2e– → Ca(s) K+(aq) ) + e– → K(s) Li+(aq) + e– → Li(s)
Note that NO3- (aq) cannot be oxidised under reasonable conditions
E° / V +2.01 +1.82 +1.72 +1.69 +1.69 +1.51 +1.50 +1.36 +1.23 +1.18 +1.09 +0.96 +0.96 +0.92 +0.88 +0.80 +0.80 +0.77 +0.54 +0.53 +0.40 +0.34 +0.15 0 (by definition) –0.04 –0.126 –0.136 –0.24 –0.28 –0.40 -0.43 –0.44 –0.74 –0.76 –0.83 –0.89 −1.16 –1.68 –2.36 –2.71 –2.87 –2.94 –3.04
Useful formulas Quantum Chemistry
Electrochemistry
E = h = hc/
G = –nFE
= h/mv
Moles of e– = It/F
E = –Z2ER(1/n2)
E = E – (RT/nF) lnQ
x(mv) h/4 T = 2.898 106 K nm
E = (RT/nF) lnK 0.0592 E = E – logQ (at 25 C) n
Acids and Bases
Gas Laws
pH = –log[H+]
PV = nRT
pKw = pH + pOH = 14.00
(P + n2a/V2)(V – nb) = nRT
pKw = pKa + pKb = 14.00
Ek = ½mv2
q = 4r2 5.67 10–8 T4
–
pH = pKa + log{[A ] / [HA]} Radioactivity
Kinetics
t½ = ln2/k
t½ = ln2/k
A = kN
k = Ae–Ea/RT
ln(N0/Nt) = kt
ln[A] = ln[A]0 – kt
14
C age = 8033 ln(A0/At) years
ln
k2 Ea 1 = ( - 1) k1 R T1 T 2
Colligative Properties & Solutions
Thermodynamics & Equilibrium
= cRT
G = H – TS
Psolution = Xsolvent Psolvent
G = G + RT lnQ
c = kp
G = –RT lnK
Tf = Kfm
univS = R lnK K p = Kc (RT ) n
Tb = Kbm Miscellaneous I
A = –log
I0
Mathematics
If ax2 + bx + c = 0, then x =
A = cl
ln x = 2.303 log x
2 E = –A e NA 4 0r
Area of circle = r2 Surface area of sphere = 4r2
−b
b2 − 4ac 2a
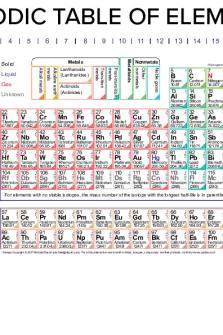
PERIODIC TABLE OF THE ELEMENTS 1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
1
2
HYDROGEN
HELIUM
H
He
1.008
4.003
3
4
6
7
8
9
LITHIUM
BERYLLIUM
BORON
5
CARBON
NITROGEN
OXYGEN
FLUORINE
10 NEON
Li
Be
B
C
N
O
F
Ne
6.941
9.012
10.81
12.01
14.01
16.00
19.00
20.18
11
12
13
14
15
16
17
18
SODIUM
MAGNESIUM
ALUMINIUM
SILICON
PHOSPHORUS
SULFUR
CHLORINE
ARGON
Na
Mg
Al
Si
P
S
Cl
Ar
22.99
24.31
26.98
28.09
30.97
32.07
35.45
39.95
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
POTASSIUM
CALCIUM
SCANDIUM
TITANIUM
VANADIUM
CHROMIUM
MANGANESE
IRON
COBALT
NICKEL
COPPER
ZINC
GALLIUM
GERMANIUM
ARSENIC
SELENIUM
BROMINE
KRYPTON
K
Ca
Sc
Ti
V
Cr
Mn
Fe
Co
Ni
Cu
Zn
Ga
Ge
As
Se
Br
Kr
39.10
40.08
44.96
47.88
50.94
52.00
54.94
55.85
58.93
58.69
63.55
65.39
69.72
72.59
74.92
78.96
79.90
83.80
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
53
54
RUBIDIUM
STRONTIUM
YTTRIUM
ZIRCONIUM
MOLYBDENUM
TECHNETIUM
RUTHENIUM
RHODIUM
PALLADIUM
SILVER
CADMIUM
INDIUM
TIN
IODINE
XENON
NIOBIUM
ANTIMONY
TELLURIUM
Rb
Sr
Y
Zr
Nb
Mo
Tc
Ru
Rh
Pd
Ag
Cd
In
Sn
Sb
Te
I
Xe
85.47
87.62
88.91
91.22
92.91
95.94
[98.91]
101.07
102.91
106.4
107.87
112.40
114.82
118.69
121.75
127.60
126.90
131.30
55
56
57-71
CAESIUM
BARIUM
72
73
74
75
76
77
78
79
80
81
82
HAFNIUM
TANTALUM
TUNGSTEN
RHENIUM
OSMIUM
IRIDIUM
PLATINUM
GOLD
MERCURY
THALLIUM
LEAD
83 BISMUTH
84
85
86
POLONIUM
ASTATINE
RADON
Cs
Ba
Hf
Ta
W
Re
Os
Ir
Pt
Au
Hg
Tl
Pb
Bi
Po
At
Rn
132.91
137.34
178.49
180.95
183.85
186.2
190.2
192.22
195.09
196.97
200.59
204.37
207.2
208.98
[210.0]
[210.0]
[222.0]
105
106
107
110
111
DUBNIUM
SEABORGIUM
BOHRIUM
87
88
FRANCIUM
89-103 104
RADIUM
RUTHERFORDIUM
108 HASSIUM
109 MEITNERIUM
DARMSTADTIUM ROENTGENIUM
112
113
COPERNICIUM
NIHONIUM
114 FLEROVIUM
115
116
117
MOSCOVIUM
LIVERMORIUM
TENNESSINE
118 OGANESSON
Fr
Ra
Rf
Db
Sg
Bh
Hs
Mt
Ds
Rg
Cn
Nh
Fl
Mc
Lv
Ts
Og
[223.0]
[226.0]
[263]
[268]
[271]
[274]
[270]
[278]
[281]
[281]
[285]
[286]
[289]
[290]
[293]
[294]
[294]
57 LANTHANOIDS
ACTINOIDS
LANTHANUM
58
59
60
61
62
CERIUM
PRASEODYMIUM
NEODYMIUM
PROMETHIUM
SAMARIUM
63 EUROPIUM
64 GADOLINIUM
65 TERBIUM
66 DYSPROSIUM
67 HOLMIUM
68 ERBIUM
69 THULIUM
70 YTTERBIUM
71 LUTETIUM
La
Ce
Pr
Nd
Pm
Sm
Eu
Gd
Tb
Dy
Ho
Er
Tm
Yb
Lu
138.91
140.12
140.91
144.24
[144.9]
150.4
151.96
157.25
158.93
162.50
164.93
167.26
168.93
173.04
174.97
89
90
91
92
93
94
95
96
ACTINIUM
THORIUM
PROTACTINIUM
URANIUM
NEPTUNIUM
PLUTONIUM
AMERICIUM
CURIUM
97 BERKELLIUM
98
99
100
101
102
103
CALIFORNIUM
EINSTEINIUM
FERMIUM
MENDELEVIUM
NOBELIUM
LAWRENCIUM
Ac
Th
Pa
U
Np
Pu
Am
Cm
Bk
Cf
Es
Fm
Md
No
Lr
[227.0]
232.04
[231.0]
238.03
[237.0]
[239.1]
[243.1]
[247.1]
[247.1]
[252.1]
[252.1]
[257.1]
[256.1]
[259.1]
[260.1]...
Similar Free PDFs

Datasheet and periodic table
- 4 Pages

Periodic table
- 2 Pages
Periodic-table
- 1 Pages

Formula sheet and periodic table
- 1 Pages

Periodic Table Virtual Lab
- 3 Pages

Periodic table worksheet
- 8 Pages

Iupac Periodic Table 2016
- 2 Pages

Alien Periodic Table
- 3 Pages

The Periodic Table
- 8 Pages

Periodic Table Summary Notes
- 11 Pages

History of Periodic Table
- 1 Pages

Iupac Periodic Table
- 1 Pages

Periodic Table Packet
- 4 Pages

Periodic Table & Periodicity
- 9 Pages
Popular Institutions
- Tinajero National High School - Annex
- Politeknik Caltex Riau
- Yokohama City University
- SGT University
- University of Al-Qadisiyah
- Divine Word College of Vigan
- Techniek College Rotterdam
- Universidade de Santiago
- Universiti Teknologi MARA Cawangan Johor Kampus Pasir Gudang
- Poltekkes Kemenkes Yogyakarta
- Baguio City National High School
- Colegio san marcos
- preparatoria uno
- Centro de Bachillerato Tecnológico Industrial y de Servicios No. 107
- Dalian Maritime University
- Quang Trung Secondary School
- Colegio Tecnológico en Informática
- Corporación Regional de Educación Superior
- Grupo CEDVA
- Dar Al Uloom University
- Centro de Estudios Preuniversitarios de la Universidad Nacional de Ingeniería
- 上智大学
- Aakash International School, Nuna Majara
- San Felipe Neri Catholic School
- Kang Chiao International School - New Taipei City
- Misamis Occidental National High School
- Institución Educativa Escuela Normal Juan Ladrilleros
- Kolehiyo ng Pantukan
- Batanes State College
- Instituto Continental
- Sekolah Menengah Kejuruan Kesehatan Kaltara (Tarakan)
- Colegio de La Inmaculada Concepcion - Cebu

