Determination of Mn in Steel via Spectrophotometry new PDF

| Title | Determination of Mn in Steel via Spectrophotometry new |
|---|---|
| Course | General Chemistry I |
| Institution | Hofstra University |
| Pages | 4 |
| File Size | 184.7 KB |
| File Type | |
| Total Downloads | 98 |
| Total Views | 180 |
Summary
Download Determination of Mn in Steel via Spectrophotometry new PDF
Description
Determination of Mn in Steel via Spectrophotometry Olivia pizzella Oct 4th 2020 Dr.Nirode
Mass of standard sample = 0.5174g, mass of unknown standard sample = 0.4198g wavelength : N/A (scan not in email) Calibration plot
The standard is 0.83% Mn by mass Absorbance & concentrations for Mn in the five standard samples Concentration
Absorbance 0
0.476
0.002257
0.124
0.003695
0.203
0.005461
0.3
0.007227
0.397
Absorbance and calculator Mn concentration in unknown samples ( unknown N/A was not sent in email) Unknown is 97.29% Absorbance & concentrations for Mn in the five standard samples unknown concentration
absorbance 0.0091556
0.382
0.0020753 0.0028217 0.0046422 0.0055889
0.114 0.155 0.255 0.307
Summary
Unknown #
N/A not sent in email
Average
0.00485674
Standard std
0.002487572
95% conf. Interval
0.004857 ±0.00218
Calculations: 1. Dilutions of the standard sample (and of unknown if different) ■ m1= 0.1910 2. Sample calculation for Mn concentration in diluted standard (used to create plot) ■ x=0.000000019047619 3. Sample calculation for Mn concentration (as %) in unknown sample ■ 0.0206%
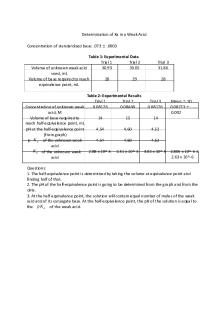
Thought Questions: 1.Why is phosphoric acid added to the Mn steel solution? Ferric ions are produced from the iron that can be major interferences in the determination of the magnanes, to prevent this we add phosphate 2.Chromium is also found in steel at trace levels. If present, the chromium would have been oxidized to chromate or dichromate. Both chromate and dichromate absorb light
in the visible region and could thus have interfered with your Mn measurements. How would standard addition calibration have corrected for this interference? -
the standard sample minimizes the spectral interferences in the analysis...
Similar Free PDFs

Heat Treatment of Steel
- 20 Pages

Types of Steel Columns
- 1 Pages

Design of Steel Structures
- 179 Pages

Determination of Ksp CuIO3
- 5 Pages

Experimental Determination of π
- 3 Pages

Iodometric Determination of Copper
- 13 Pages

Coefficient of Determination
- 2 Pages
Popular Institutions
- Tinajero National High School - Annex
- Politeknik Caltex Riau
- Yokohama City University
- SGT University
- University of Al-Qadisiyah
- Divine Word College of Vigan
- Techniek College Rotterdam
- Universidade de Santiago
- Universiti Teknologi MARA Cawangan Johor Kampus Pasir Gudang
- Poltekkes Kemenkes Yogyakarta
- Baguio City National High School
- Colegio san marcos
- preparatoria uno
- Centro de Bachillerato Tecnológico Industrial y de Servicios No. 107
- Dalian Maritime University
- Quang Trung Secondary School
- Colegio Tecnológico en Informática
- Corporación Regional de Educación Superior
- Grupo CEDVA
- Dar Al Uloom University
- Centro de Estudios Preuniversitarios de la Universidad Nacional de Ingeniería
- 上智大学
- Aakash International School, Nuna Majara
- San Felipe Neri Catholic School
- Kang Chiao International School - New Taipei City
- Misamis Occidental National High School
- Institución Educativa Escuela Normal Juan Ladrilleros
- Kolehiyo ng Pantukan
- Batanes State College
- Instituto Continental
- Sekolah Menengah Kejuruan Kesehatan Kaltara (Tarakan)
- Colegio de La Inmaculada Concepcion - Cebu