Determination of the Acid Ionization Constant PDF

| Title | Determination of the Acid Ionization Constant |
|---|---|
| Course | (Uc) General Chemistry Ii Lab |
| Institution | Quinnipiac University |
| Pages | 3 |
| File Size | 80.4 KB |
| File Type | |
| Total Downloads | 51 |
| Total Views | 169 |
Summary
Lab Report...
Description
Determination of the Acid Ionization Constant, Ka
Purpose The purpose of this experiment is to determine the pKa of a weak monoprotic acid. Procedure Given in Course Material File Data Tables None Calculations ½ way point pH = pKa
pKa = -log (Ka)
Ka = 10 ^(-pH)
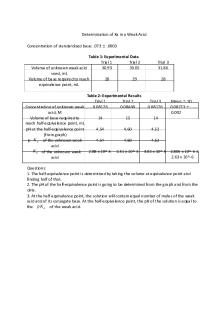
1. pH = pKa = 4.63 2. Ka= 10(-4.63) 3. Ka = 2.344 x 10 - 5 Summary of Results
Equivalence Point Volume: 21.50 mL
Half Equivalence Point Volume: 10.75 mL
pKa: 4.63
Ka: 2.344 x 10 – 5
Conclusion The purpose of this lab was to determine the pKa of a weak monoprotic acid, and this was accomplished by the calculations shown above. The equivalence point was found by determining where the volume that corresponds to the highest peak in the plot was for the first derivative plot, which was 21.50 mL. The half equivalence point volume was determined by diving the equivalence point volume in half with a value of 0.75 mL. The pH is equal to the pKa at half the equivalence volume, this was determined to be 4.63. The ka value was calculated by raising 10 to the -4.63 power, concluding a Ka value of 2.344 x 10 – 5. Errors that may have occurred in this lab is if the volume was not recorded correctly from the buret. Another error that may have occurred
while conducting this experiment is if is calibration of the pH electrode was not done properly. If calibration is off then pH is offset high or low....
Similar Free PDFs

Acid Ionization Lab Report
- 8 Pages
Popular Institutions
- Tinajero National High School - Annex
- Politeknik Caltex Riau
- Yokohama City University
- SGT University
- University of Al-Qadisiyah
- Divine Word College of Vigan
- Techniek College Rotterdam
- Universidade de Santiago
- Universiti Teknologi MARA Cawangan Johor Kampus Pasir Gudang
- Poltekkes Kemenkes Yogyakarta
- Baguio City National High School
- Colegio san marcos
- preparatoria uno
- Centro de Bachillerato Tecnológico Industrial y de Servicios No. 107
- Dalian Maritime University
- Quang Trung Secondary School
- Colegio Tecnológico en Informática
- Corporación Regional de Educación Superior
- Grupo CEDVA
- Dar Al Uloom University
- Centro de Estudios Preuniversitarios de la Universidad Nacional de Ingeniería
- 上智大学
- Aakash International School, Nuna Majara
- San Felipe Neri Catholic School
- Kang Chiao International School - New Taipei City
- Misamis Occidental National High School
- Institución Educativa Escuela Normal Juan Ladrilleros
- Kolehiyo ng Pantukan
- Batanes State College
- Instituto Continental
- Sekolah Menengah Kejuruan Kesehatan Kaltara (Tarakan)
- Colegio de La Inmaculada Concepcion - Cebu