CU Su20 Determination of Acetic Acid Concentration Q PDF

| Title | CU Su20 Determination of Acetic Acid Concentration Q |
|---|---|
| Author | Alison Naeve |
| Course | Laboratory in General Chemistry 1 |
| Institution | University of Colorado Boulder |
| Pages | 10 |
| File Size | 324.1 KB |
| File Type | |
| Total Downloads | 91 |
| Total Views | 142 |
Summary
LAB WORK...
Description
Determination of Acetic Acid Concentration in Vinegar Using Titration Student Name: Alison Naeve Date: 10/15/2020
1
Introduction What is being determined in this lab? Please provide a brief (2-3 sentences) answer in your own words. The purpose of the lab was to accurately determine the concentration of acetic acid. Activity 1 uses vinegar with a determined amount of acetic acid while activity 2 uses a different sample of vinegar to be titrated.
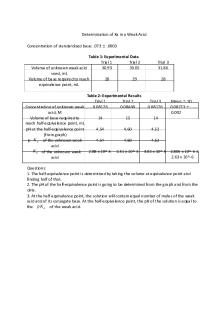
Data Activity 1 Data Table 1 Vinegar Sample 1
Vinegar Sample 2
Vinegar Sample 3
5.03
5.01
4.97
1 g=1 mL 5.03 g=5.03 mL
5.01
4.97
Initial NaOH Volume in Syringe
10.0
10.0
9.9
Final NaOH Volume in Syringe
6.0
5.9
5.7
Name of Vinegar Sample
Mass of Vinegar
Volume of Vinegar
© 2016 Carolina Biological Supply Company
2
Name of Vinegar Sample
Vinegar Sample 1
Vinegar Sample 2
Vinegar Sample 3
Volume NaOH Delivered
10-6= 4.0
4.1
4.3
Moles of NaOH Delivered
0.10
0.10
0.10
Moles of Acetic Acid in Sample
0.10
0.10
0.11
Mass of Acetic Acid in Sample
6.0
6.0
6.6
Calculated Molarity of Acetic Acid in Sample Vinegar
20.0
20.0
22
Calculated Percent Mass of Acetic Acid in Sample Vinegar
54.4%
54.5%
51.8%
Average Calculated Percent Mass of Acetic Acid in Sample Vinegar
53.6%
53.6%
53.6%
© 2016 Carolina Biological Supply Company
3 CH3CO2H+ NaOH- NaCH3CO2+H2O 0.10 mol NaOH * 1 mol CH3CO2H/ 1 mol NaOH = 0.10 mol CH3CO2H 0.10 mol CH3CO2H * 60.052 g CH3CO2H/ 1 mol CH3CO2H= 6.0 g CH3CO2H (0.10 mol CH3CO2H)/ 0.005 L= 20.0 M (Percent mass) (6.0 g/ (5.03 g + 6.0 g)) * 100= 54.4% (average Percent mass) 54.4+54.5 +51.8)/300= 53.6%
Activity 2
Data Table 2 Vinegar Sample 1
Vinegar Sample 2
Vinegar Sample 3
5.02
5.08
4.97
1 g= 1mL 5.02 g= 5.02 mL
5.08
4.97
Initial NaOH Volume in Syringe
9.8
10
9.9
Final NaOH Volume in Syringe
7.6
7.2
7.1
Volume NaOH Delivered
2.2
2.8
2.8
Name of Vinegar Sample
Mass of Vinegar
Volume of Vinegar
© 2016 Carolina Biological Supply Company
4 Vinegar Sample 1
Vinegar Sample 2
Vinegar Sample 3
Moles of NaOH Delivered
0.055
0.070
0.070
Moles of Acetic Acid in Sample
0.055
0.070
0.070
Mass of Acetic Acid in Sample
3.3
4.2
4.2
Calculated Molarity of Acetic Acid in Sample Vinegar
11
14
14
Calculated Percent Mass of Acetic Acid in Sample Vinegar
39.7%
45.3%
45.8%
Average Calculated Percent Mass of Acetic Acid in Sample Vinegar
43.5%
43.5%
43.5%
Name of Vinegar Sample
% CH3CO2H+NaOH NaCH3CO2+H2O 0.055 mol NaOH * 1 mol CH3CO2H/ 1 mol NaOH+ 0.055 mol CH3CO2H 0.055 mol CH3CO2H* 60.052g CH3CO2H/ 1 mol CH3CO2H= 3.3 g CH3CO2H (0.055 mol CH3CO2H)/ 0.00502= 11 M (Percent mass)- (3.3g/ (3.3g +5.02g)) * 100= 54.4% (Average percent mass)- (39.4+45.3+45.8)/ 300= 43.5% 1. List at least two possible sources of error from the experiment. Would these errors result in an overestimation or an underestimation of the concentration of acetic acid in the vinegar?
© 2016 Carolina Biological Supply Company
5 Two possible sources of error were the plunger being pushed too hard whi ch would release too much NaOH into the vinegar at once or the error of the scale which could be in not giving an accurate reading. The first source of error could be a result from overestimating the concentration of acetic acid in vinegar. All of these things would result in off or different answers/results.
Photos
Photo 1 Insert the photo of the 25-mL Erlenmeyer flask after the addition of phenolphthalein from Activity 1, step 6.
© 2016 Carolina Biological Supply Company
6
© 2016 Carolina Biological Supply Company
7
Photo 2 Insert the photo of the 25-mL Erlenmeyer flask at the endpoint of the titration from Activity 1, step 13.
© 2016 Carolina Biological Supply Company
8
© 2016 Carolina Biological Supply Company
9
Conclusion Write a one paragraph (3-5 sentences) statement that provides the scientific objective of the lab, a brief summary of the procedure, and report the major results. The purpose of the lab was to accuratly determine the concentration of acetic acid which we did in activy 1 as well as finding the average calucated percent mass of Acetic acid in a sample (53.6%). In activty 2 a different sample of vinegar yeiled an average percent mass of 43.5%. The concentration of the acetic acid in the sample used in activity 2 was and should be lower than the first was the samples were the same but flushed in the second ecpierament helpign prove our results.
Be sure to show your calculations (at least one sample calculation for every calculated step) by either typing in the calculation under the data table or adding a picture of calculations done by hand!
© 2016 Carolina Biological Supply Company...
Similar Free PDFs
Popular Institutions
- Tinajero National High School - Annex
- Politeknik Caltex Riau
- Yokohama City University
- SGT University
- University of Al-Qadisiyah
- Divine Word College of Vigan
- Techniek College Rotterdam
- Universidade de Santiago
- Universiti Teknologi MARA Cawangan Johor Kampus Pasir Gudang
- Poltekkes Kemenkes Yogyakarta
- Baguio City National High School
- Colegio san marcos
- preparatoria uno
- Centro de Bachillerato Tecnológico Industrial y de Servicios No. 107
- Dalian Maritime University
- Quang Trung Secondary School
- Colegio Tecnológico en Informática
- Corporación Regional de Educación Superior
- Grupo CEDVA
- Dar Al Uloom University
- Centro de Estudios Preuniversitarios de la Universidad Nacional de Ingeniería
- 上智大学
- Aakash International School, Nuna Majara
- San Felipe Neri Catholic School
- Kang Chiao International School - New Taipei City
- Misamis Occidental National High School
- Institución Educativa Escuela Normal Juan Ladrilleros
- Kolehiyo ng Pantukan
- Batanes State College
- Instituto Continental
- Sekolah Menengah Kejuruan Kesehatan Kaltara (Tarakan)
- Colegio de La Inmaculada Concepcion - Cebu