04 - Qualitative Analysis of Solutions PDF

| Title | 04 - Qualitative Analysis of Solutions |
|---|---|
| Author | M. T. |
| Course | Chemistry |
| Institution | High School - Canada |
| Pages | 2 |
| File Size | 66.2 KB |
| File Type | |
| Total Downloads | 51 |
| Total Views | 158 |
Summary
Notes...
Description
4 - Qualitative Analysis of Solutions
SCH3UE – Solution Chemistry
Date: ______________________________________________
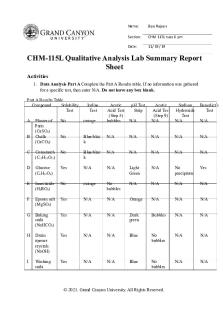
Qualitative Analysis of Solutions A solution may contain one or all of the following ions: Ag+, Ba2+, and Mg2+ Using your understanding of the solubility rules and chemical reactions, devise a test that you could use to identify which of these ions are present in the solution Use net ionic equations to illustrate a positive test for each ion
Choose an anion to precipitate only silver ions
Choose an anion to precipitate only barium but not magnesium ions
Choose an anion to precipitate only magnesium ions
Suppose that the solution contain all of these ions Prepare a procedure that could be used to selectively separate each of these ions from each other and the solution
Page 1 of 2
4 - Qualitative Analysis of Solutions
SCH3UE – Solution Chemistry
Solution Colour The identity of a metal ion can be inferred by the colour of aqueous solution it forms Ion
Solution Colour
Cu2+
_________________________
Co2+
_________________________
Ni2+
_________________________
MnO4-
_________________________
Alkali Metals
_________________________
Flame Test The identity of a metal (atom or ion) can be inferred by the colour of the flame containing the metal Metal
Flame colour
Li+
_________________________
Na+
_________________________
K+
_________________________
Cu+
_________________________
Cu2+
_________________________
Sr2+
_________________________
Page 2 of 2...
Similar Free PDFs

Qualitative Analysis of Anions
- 11 Pages

Qualitative Analysis of Carbohydrate
- 13 Pages

Qualitative Analysis of Cations
- 27 Pages

Exam Qualitative analysis sample
- 4 Pages

Task 1 - Qualitative analysis
- 4 Pages

Qualitative Structural Analysis
- 18 Pages

Qualitative Analysis - Beams
- 15 Pages

Qualitative Analysis-Sp21
- 6 Pages

5. Qualitative Movement Analysis
- 1 Pages
Popular Institutions
- Tinajero National High School - Annex
- Politeknik Caltex Riau
- Yokohama City University
- SGT University
- University of Al-Qadisiyah
- Divine Word College of Vigan
- Techniek College Rotterdam
- Universidade de Santiago
- Universiti Teknologi MARA Cawangan Johor Kampus Pasir Gudang
- Poltekkes Kemenkes Yogyakarta
- Baguio City National High School
- Colegio san marcos
- preparatoria uno
- Centro de Bachillerato Tecnológico Industrial y de Servicios No. 107
- Dalian Maritime University
- Quang Trung Secondary School
- Colegio Tecnológico en Informática
- Corporación Regional de Educación Superior
- Grupo CEDVA
- Dar Al Uloom University
- Centro de Estudios Preuniversitarios de la Universidad Nacional de Ingeniería
- 上智大学
- Aakash International School, Nuna Majara
- San Felipe Neri Catholic School
- Kang Chiao International School - New Taipei City
- Misamis Occidental National High School
- Institución Educativa Escuela Normal Juan Ladrilleros
- Kolehiyo ng Pantukan
- Batanes State College
- Instituto Continental
- Sekolah Menengah Kejuruan Kesehatan Kaltara (Tarakan)
- Colegio de La Inmaculada Concepcion - Cebu