Solubility Flowchart PDF

| Title | Solubility Flowchart |
|---|---|
| Course | Chemistry I |
| Institution | University of Ontario Institute of Technology |
| Pages | 2 |
| File Size | 79.3 KB |
| File Type | |
| Total Downloads | 99 |
| Total Views | 170 |
Summary
Solubility flowchart...
Description
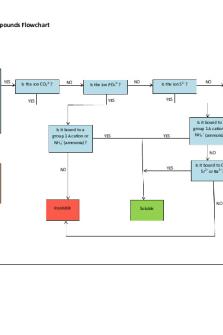
Insoluble Compounds Flowchart
Does the compound contain one of the following ions? CO3 2- (carbonate) S2- (sulfide) PO43- (phosphate) OH- (hydroxide)
YES
NO
Is the ion CO3 2- ?
Is the ion PO4 3- ?
NO
Is the ion S2- ?
Is it bound to a group 1 A cation or NH4+ (ammonia)?
Is the ion OH- ?
YES
YES
YES
NO
YES
YES
YES
Is it bound to a group 1 A cation or NH4 + (ammonia)?
NO May be soluble – consider reviewing the soluble compounds flowchart
YES
NO
Is it bound to Ca 2+, Sr2+ or Ba2+?
NO Insoluble
Soluble
Soluble Compounds Flowchart
Is the ion Cl-, Br - or I- ?
YES
NO
YES
NO
Is the ion SO42- ?
YES Soluble
Does the compound contain one of the following ions:
Is the ion Cl -, Br- or Ibound to Ag+ Hg2+ or Pb2+? NO
Li+ Na+ K+ Rb+ Cs+ (group 1A cations) NH4 + (ammonium ion)
Is SO42- bound to Sr 2+, Ba2+ Hg2+ or Pb2+ Ca2+?
YES
Cl- Br- I-
YES
NO3- (Nitrate) ClO4 (Perchlorate) CH3CO2 (Acetate) SO4 2- (sulfate) ClO3 - (Chlorate) NO Insoluble
NO...
Similar Free PDFs
Solubility Flowchart
- 2 Pages

27 Solubility-S - Solubility
- 4 Pages

Flowchart-acco - flowchart
- 1 Pages

Flowchart
- 8 Pages
Flowchart
- 1 Pages

Solubility Guidelines
- 1 Pages

Solubility Rules
- 1 Pages

Predicting Solubility
- 1 Pages

Solubility Rules
- 1 Pages

Solubility Rules
- 1 Pages

Solubility Table
- 1 Pages

Solubility Equilibrium
- 8 Pages

Solubility Product Lab
- 5 Pages

Negligence Flowchart
- 3 Pages
Popular Institutions
- Tinajero National High School - Annex
- Politeknik Caltex Riau
- Yokohama City University
- SGT University
- University of Al-Qadisiyah
- Divine Word College of Vigan
- Techniek College Rotterdam
- Universidade de Santiago
- Universiti Teknologi MARA Cawangan Johor Kampus Pasir Gudang
- Poltekkes Kemenkes Yogyakarta
- Baguio City National High School
- Colegio san marcos
- preparatoria uno
- Centro de Bachillerato Tecnológico Industrial y de Servicios No. 107
- Dalian Maritime University
- Quang Trung Secondary School
- Colegio Tecnológico en Informática
- Corporación Regional de Educación Superior
- Grupo CEDVA
- Dar Al Uloom University
- Centro de Estudios Preuniversitarios de la Universidad Nacional de Ingeniería
- 上智大学
- Aakash International School, Nuna Majara
- San Felipe Neri Catholic School
- Kang Chiao International School - New Taipei City
- Misamis Occidental National High School
- Institución Educativa Escuela Normal Juan Ladrilleros
- Kolehiyo ng Pantukan
- Batanes State College
- Instituto Continental
- Sekolah Menengah Kejuruan Kesehatan Kaltara (Tarakan)
- Colegio de La Inmaculada Concepcion - Cebu